© Copyright 2002-2026, Generis Enterprise Technology Limited, all rights reserved
Your leadership wants AI. Your compliance team wants governance.
Your team still does it manually.
A secure, validated AI layer inside the CARA Platform for R&D, Regulatory, Safety and Quality teams. Keep data in-platform, automate & connect processes across the enterprise.








THE PROBLEM WITH AI IN PHARMA
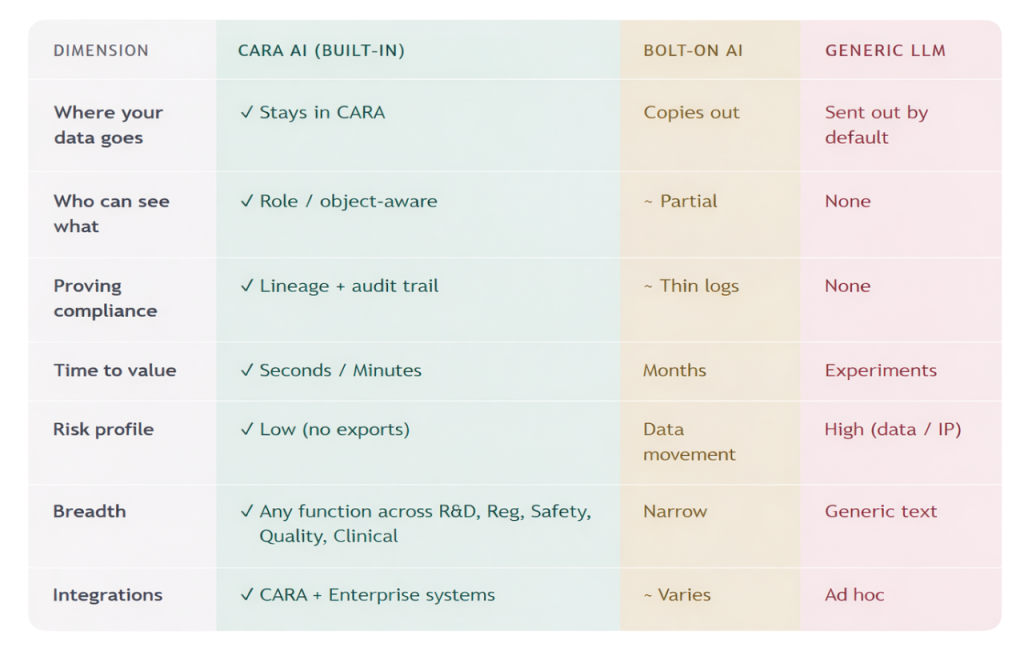
Here’s what changes when AI is built into a unified platform
not bolted on top of fragmented data.
With CARA AI
Every output is source-linked and audit-ready.
Full traceability to controlled, validated content. Defensible in any inspection, by design.
One foundation. Cross-functional intelligence.
All functions share one data model. The AI draws from all of them in a single pass natively.
GxP compliance is the architecture.
Not a feature added on top. The platform was built for regulated industries from the ground up.
Fragmented systems don’t just create manual work. They limit what AI can do. Every system you add creates costs you didn’t budget for.

The comparison isn’t about features.
It’s about what the architecture makes possible and what it makes impossible.
One governed AI layer across Regulatory, Safety, Quality, and Clinical,
full traceability on every output.
Context-aware search
Surfaces regulatory precedents and clinical correlations that keyword search misses.
Automated classification
Every document tagged, categorised, and enriched automatically. Instant retrieval, every time.
Pre-tuned for compliance
Built-in prompts for regulatory workflows. Accurate from day one, customisable further.
Your LLM
Choose the model that fits. All processing stays in our secure framework. Your IP stays yours.
Data extraction
Pulls structured data from messy PDFs and clinical reports. Analysis-ready in seconds.
No separate system. No separate contract. No separate validation cycle.
Every function Regulatory, Safety, Quality, Clinical, delivers value from day one.
And the more of your operations run on CARA, the more your AI can see, connect, and do.
PBRER highlights without reading 60 pages
Audit reports distilled to what actually matters
100 pages of trial data — clear insights today
Variation narratives and submission documents — not from scratch
PBRER first drafts and RMP annexes from live data
SOPs and CAPAs — first drafts in minutes, not days
Patient narratives and CSR sections — consistently formatted
Label changes checked against your PSMF, RMP, and submission history in one pass
Signal trends across your portfolio — spotted, not missed
Protocol deviations across sites — at a glance
Health Authority query patterns — anticipated before they arrive
Benefit-risk signals surfaced before reporting deadlines
Recurring deviations flagged before they compound
Data lock risks visible weeks before they delay timelines
Submission dossiers in local formats — without starting over
RMP annexes for multiple markets — technically consistent
SOPs across countries — technically accurate, no rework
ICFs in multiple languages — medically accurate across markets
Spend your time driving results, not drowning in documentation
Lower audit exposure with provable lineage
Faster cycles across teams; fewer handoffs
Lower cost per change/submission; fewer surprises
PBRER highlights without reading 60 pages
Audit reports distilled to what actually matters
100 pages of trial data — clear insights today
Variation narratives and submission documents — not from scratch
PBRER first drafts and RMP annexes from live data
SOPs and CAPAs — first drafts in minutes, not days
Patient narratives and CSR sections — consistently formatted
Label changes checked against your PSMF, RMP, and submission history in one pass
Signal trends across your portfolio — spotted, not missed
Protocol deviations across sites — at a glance
Health Authority query patterns — anticipated before they arrive
Benefit-risk signals surfaced before reporting deadlines
Recurring deviations flagged before they compound
Data lock risks visible weeks before they delay timelines
Submission dossiers in local formats — without starting over
RMP annexes for multiple markets — technically consistent
SOPs across countries — technically accurate, no rework
ICFs in multiple languages — medically accurate across markets
Spend your time driving results, not drowning in documentation
Lower audit exposure with provable lineage
Faster cycles across teams; fewer handoffs
Lower cost per change/submission; fewer surprises
Each function has a dedicated page with deeper use cases, implementation detail,
and outcome evidence.
HOW TO GET STARTED
Deploy CARA AI for one function today. When a second function is ready, they join the same platform no new vendor, no new validation, no new RFP process.
Potential Objection
A label comparison that also checks safety signals. A CAPA that surfaces related deviations across sites. A submission that draws from clinical, regulatory, and quality content simultaneously. These use cases only exist on a unified platform.
Regulatory, Safety, Quality, or Clinical — each delivers immediate value on its own from day one.
Same platform. Same validation. Same vendor. A second function joins the foundation — it doesn’t integrate with it. No new RFP required.
The more functions on the platform, the more your AI can do — with use cases that simply don’t exist on any other architecture.
A label comparison that also checks safety signals. A CAPA that surfaces related deviations across sites. A submission that draws from clinical, regulatory, and quality content simultaneously. These use cases only exist on a unified platform.
Widely regarded by our customers for our responsiveness, we offer unprecedented care and attention to deliver a high-performing platform for all your needs. CARA now counts over 1,000,000 users across industries worldwide, including 8 of the 10 largest Life Science companies.







60%
of top pharma
1M+
Users
99.9%
Uptime SLA
Built on a platform that enterprise pharma already trusts
CARA AI runs on the same unified foundation already deployed across the world’s leading pharmaceutical companies. The platform that makes this AI possible has been proven in production, at scale, under inspection, across every major life sciences function.
25 +
Years in regulated life sciences
24/7
Expert Support
© Copyright 2002-2026, Generis Enterprise Technology Limited, all rights reserved