© Copyright 2002-2026, Generis Enterprise Technology Limited, all rights reserved
Menu
—— FOR CLINICAL TEAMS
CARA manages your full Clinical function on one governed platform, CTMS, eTMF, eTMF Archive, and Medical Writing. Add CARA AI when you’re ready. CSR and synopsis writing effort reduced by 50–70%, patient narratives auto-drafted, and protocol consistency checked automatically.
—— THE PROBLEM
Protocol amendments, label updates, deviation reports — each one creates a reconciliation cascade across documents that don’t update themselves. Expert clinical time goes on checking alignment, not running the trial.
Clinical Ops Lead · Medical Writer · eTMF Manager
Most AI tools sit on top of that same fragmented foundation. Your team still has to chase the source, reconcile versions, and govern outputs separately. You’ve added a tool. You haven’t removed the problem.
CARA AI works from inside your clinical data not on top of it. The reconciliation work disappears. Your team gets back to the trial.

Manual drafting → Auto generated
Days→ Automatic
Reactive review → Early intervention
One governed AI layer across Regulatory, Safety, Quality, and Clinical, full traceability on every output.
Finds regulatory precedents and clinical correlations keyword search misses. Better decisions, complete information.
Every document tagged, categorised, and enriched automatically. Instant retrieval, every time.
Pulls structured data from messy PDFs and clinical reports. Analysis-ready outputs in seconds.
Built-in prompts for regulatory workflows. Accurate results from day one, or customise further
Choose the model that fits your needs. All processing stays in our secure framework. Your IP stays yours.
Every output references the exact platform record it drew from.
Every output references the exact platform record it drew from.
Every output references the exact platform record it drew from.
CARA AI runs on one platform. One validation scope.The AI capabilities extend what the platform already does,
no separate system, no separate contract, no separate validation cycle.
The more of your operations run on CARA, the more your AI can do. Regulatory, Safety, Quality, and Clinical on one platform
means AI that works across your entire function not just in silos.
60%
of top pharma
1M+
Users
99.9%
Uptime SLA
Built on a platform that enterprise pharma already trusts
CARA AI runs on the same unified foundation already deployed across the world’s leading pharmaceutical companies. The platform that makes this AI possible has been proven in production, at scale, under inspection, across every major life sciences function.
25 +
Years in regulated life sciences
24/7
Expert Support
— PLATFORM ARCHITECTURE
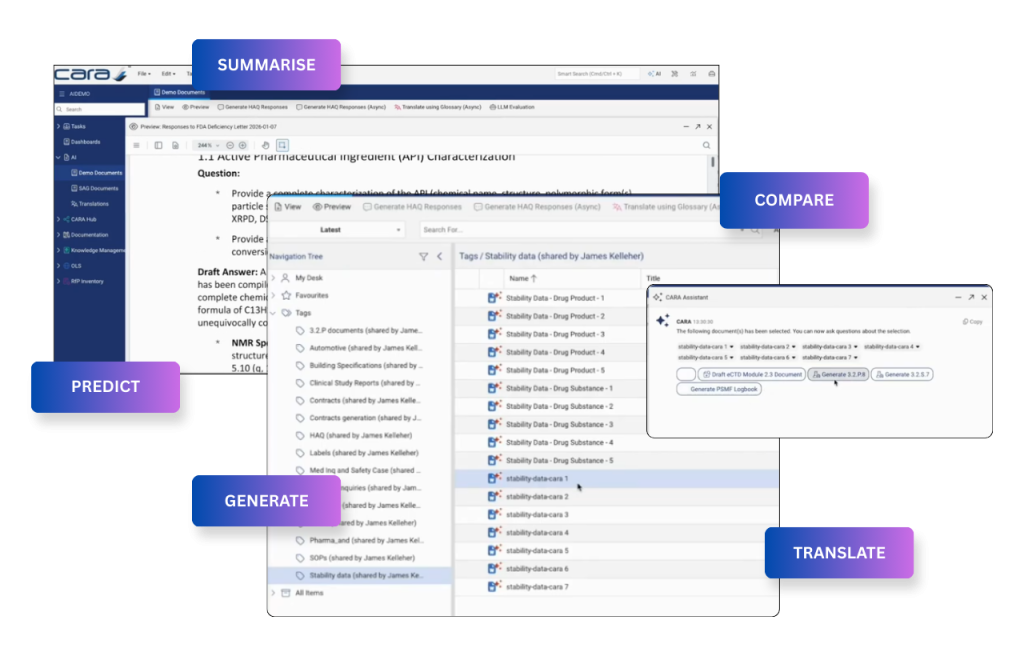
Bolt‑on AI generates content in isolation. CARA AI generates it from your live, governed regulatory content with a complete audit trail, all within your current governance framework, without extra tools, exports, or revalidation.
HA Q&A answered in minutes — not days
A Health Authority query draws on live submission records, Safety data, and Quality records in a single response. No cross-system data pull. No manual reconciliation. One platform, one governed layer.
Metric: 2–5 days → 20 minutes
Label beneath metric: Based on internal benchmarks

© Copyright 2002-2026, Generis Enterprise Technology Limited, all rights reserved