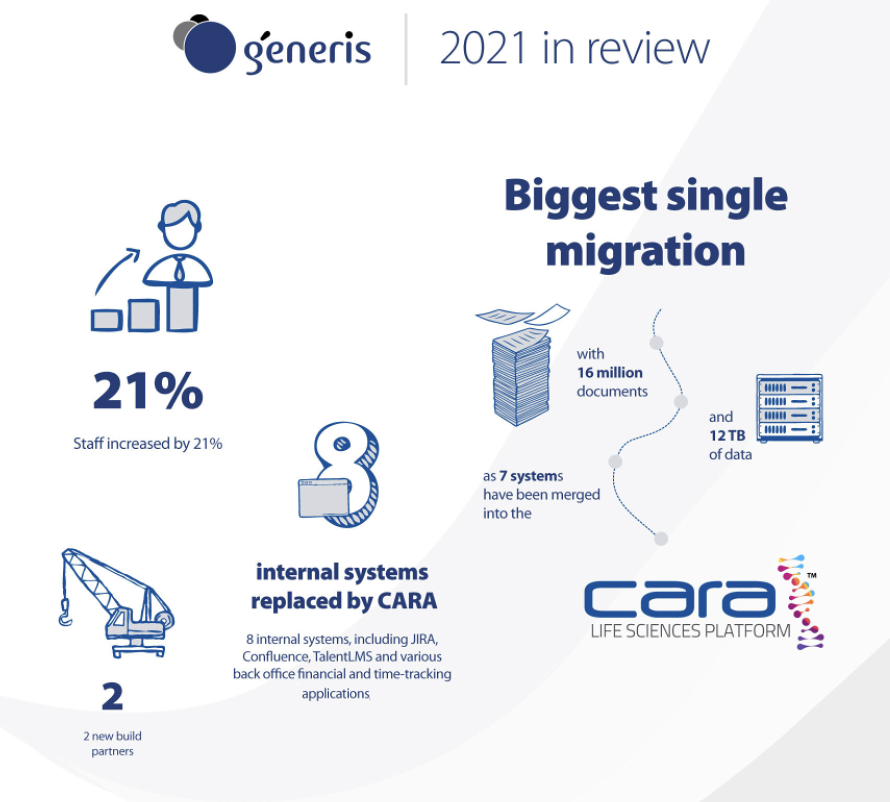
If 2020 was a bumper year for Generis, then 2021 was stronger still. Revenues rose by more than 30% and we grew our workforce by over 20%.
It’s been the year that we formally released our new branding for our end-to-end CARA Life Sciences platform, and a new trend took off among our client base – expanding the use of CARA across multiple business areas, benefitting from the “One User One Licence” model that mirrors the “enter data once and re-use everywhere” approach of linking enterprise information seamlessly. Some started with Regulatory information management and extended into Quality or Safety; others are doing the same in reverse. Once they’ve experienced the transformed usability and visibility across processes, content and data, via our cloud-based platform, they’ve identified ample opportunity to replicate the benefits and expand their operational visibility and actionable insights.

Record client wins
We have welcomed important new clients and partners during the year, from Scholl Wellness which has adopted CARA to improve Quality management and for whom we achieved a record 1 month signature-to-go live with a GxP / validated system, to Fannie Mae (the Federal National Mortgage Association in the US), which was a major migration away from Documentum to CARA, involving 16 million documents and 12 Tb of data, as 7 systems were merged onto our single platform in the cloud.
The number of new customers was more than double 2020, and extensions of use at existing customers have taken us close to 750,000 users of our software.
Japanese fluency
Our growth this year has been truly international, too. Japan is proving a particular hotbed of activity currently, as more organisations there put aside paper-based processes and embrace process digitalisation.
Major wins in the country recently have included the pharma division of Japan Tobacco, Japanese pharma and biotech group Kyowa Kirin, which is among the 40 largest pharma and biotech organisations in the world by revenue. It chose CARA to manage Health Authority correspondence, including leveraging the real-time translation capabilities to provide a multi-lingual searchable database of question and answers – helping to reduce the time taken to respond to similar questions in different languages / markets globally, and ensure consistency of responses. These new customers join our existing customers Otsuka, Ono, Taisho and Chugai – who have kept us busy with extensions of their CARA platform to Quality, Regulatory and Clinical.
A platform for development partners
Software development partners continue to embrace CARA as the platform on which to build their own offerings for their clients, providing interlinked process and data/content management capabilities instead of standalone applications. CARA’s ease of use and intuitive cloud-based ‘composability’ make it a no-brainer for any software company thinking outside of the box. Fme and Extedo both moved on providing offerings based on the CARA platform, and discussions are in progress with 2 large vendors in Quality and Regulatory respectively.
Intilaris was among the strategic partners we announced during the year, along with Google which is now working with us on AI-related capabilities, as well as supporting seamless Google Cloud integration with CARA. We continue our key partnerships with:
· FontoXML for structured content authoring
· DCLabs for conversion of documents to structured data
· Syneos Health and Iperion for domain expertise in Life Sciences
· Gens & Associates and Gartner Group for thought leadership input
· Large SI partners Cognizant, Accenture, Deloitte and TCS
· Specialist partners Main5, Inconsult, OSTHUS, Daelight Solutions and SyncWork
Bridging the disconnect
The common thread running through much of the above activity is the desire to overcome the process and data/content disconnect that persists in so many global organisations today – despite all of the noise made about digital transformation.
Of the many advantages we offer to companies, a clear line of sight across global operations is among the most significant. One major new client, which we hope to announce formally very soon, has cited anticipated benefits as including the ability to act more swiftly if a manufacturing issue on one side of the world emerges which impacts Regulatory Affairs in another territory. Alerted early enough, those involved will be able to tip off teams managing Clinical Trials and Quality around the world so they can adjust their plans accordingly.
In fact, we are using our own platform to consolidate our own business processes. This year we have replaced 7-8 internal systems, including JIRA, Confluence, TalentLMS and various back office financial and time-tracking applications, with CARA. This is delivering us all the savings and efficiency gains we promise our clients, thanks to the ability to access and manage business process workflow, data and content all in one place.
In this digital age, there’s no longer any excuse for global businesses to be disjointed, and that’s a situation we’re working hard to resolve in regulated industries across the world. Given the demand for next waves of digital transformation as companies build back for a new normal, 2022 is already promising to be another busy year.
– James Kelleher, CEO, Generis